Search results
Search for "redox catalysis" in Full Text gives 5 result(s) in Beilstein Journal of Organic Chemistry.
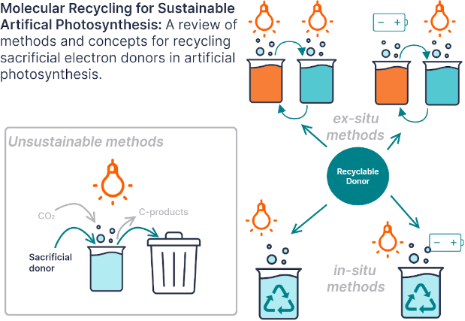
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
- establish a redox catalysis cycle [76]. In non-aqueous media DDQ has a low oxidation potential (0.14 V vs Fc/Fc+ in acetonitrile) so that DDQ could potentially reductively quench Ir(ppy)3 and Ru(bpy)3 and regenerate
Group 13 exchange and transborylation in catalysis
Beilstein J. Org. Chem. 2023, 19, 325–348, doi:10.3762/bjoc.19.28
- reactivity to be rendered catalytic, and exhibit catalysis outwith Lewis acid-type activation. These exchange reactions have allowed redox-neutral catalysis complementary to and beyond the redox catalysis of the transition metals. Boron, aluminium, gallium, and indium have all been demonstrated in catalytic
Copper catalysis with redox-active ligands
Beilstein J. Org. Chem. 2020, 16, 858–870, doi:10.3762/bjoc.16.77

- electron transfer. This review aims to present the latest results in the area of copper-based cooperative catalysis with redox-active ligands. Keywords: bioinspired catalysis; biomimetic copper complexes; cooperative catalysis; redox-active ligands; redox catalysis; Introduction Interaction of earth
- in such systems in order to develop predictive tools for reactivity control [41]. Such knowledge is most likely to result in new advances in the fast-expanding field of redox catalysis. Copper complexes with amidophenolate type benzoxazole ligands for alcohol oxidations. Copper-catalyzed aerobic
Intermolecular addition reactions of N-alkyl-N-chlorosulfonamides to unsaturated compounds
Beilstein J. Org. Chem. 2015, 11, 1226–1234, doi:10.3762/bjoc.11.136
- alkenes under copper(I) catalysis. In reactions of styrene derivatives with terminal double bonds the addition products were obtained in excellent yield and high regioselectivity. Lower yields are obtained in addition reactions to non-aromatic alkenes. The reaction most likely proceeds via a redox
- catalysis and amidyl radicals, a concerted mechanism has been ruled out and a polar mechanism via chloronium ions would lead to the opposite regiochemistry. Keywords: addition reactions; catalysis; N-chlorosulfonamides; haloamination; radical reaction; Introduction In earlier publications we described the
New developments in gold-catalyzed manipulation of inactivated alkenes
Beilstein J. Org. Chem. 2013, 9, 2586–2614, doi:10.3762/bjoc.9.294
- terminal alkenes by bimetallic gold(I) catalysis in presence of Selectfluor. Proposed mechanism for the aminoarylation of alkenes via [Au(I)-Au(I)]/[Au(II)-Au(II)] redox catalysis. Oxyarylation of terminal olefins via redox gold catalysis. a) Intramolecular gold-catalyzed oxidative coupling reactions with










































































































